Bioengineering research
Mechanical Engineering research projects
Benchtop blood flow modeling Accordion Closed
Dr. Tim Becker | Mechanical Engineering
The Bioengineering Devices Lab has developed a sophisticated comprehensive blood flow modeling system for medical device testing and surgical training.
The system models blood flow to the human brain (neurovasculature). A ViVitro programmable pump is used to simulate human blood flow from the heart and pump CMC (a blood analog developed by the BDL) throughout the system. The system consists of patient-specific vessel models, a heated reservoir, and real-time monitoring of bold pressure and flow. The current vessel model is the circle of Willis (CW) of the brain. This model features 3 inlet vessels, 6 vessel outputs. 9 pressure transducers (PTs) monitor flow throughout the CW as medical devices are introduced into the model. The benchtop flow system can operate at any blood pressure (typically set to 120/80 mmHg), blood flow rate (typically 700 mL/min to the brain via the CW), and body temperature. By changing the resistance in various regions of the flow system, any representative blood pressure and blood flow rate can be achieved and monitored in real-time. Therefore, the comprehensive flow system can be used to simulate neurovascular surgeries, test new medical devices, and train surgeons/residents on a system that better represents all aspects of the human condition.
Biomaterial designs and material characterization Accordion Closed
Dr. Tim Becker | Mechanical Engineering
The Bioengineering Devices Lab currently emphasizes three biomaterial design projects: 1) creation and mechanical testing of vessel models similar to human tissue, and 2) Modeling of delayed aneurysm rupture (DAR), and 3) innovative tissue scaffold designs using 3D-printed multi-phase surfaces
- An innovative UV cured and 3D printable acrylic-based co-polymer, VeroClear® – Agilus30® (VC-A30), is being printed and undergoing material characterization testing (compressive, tensile, and shear moduli, compliance, lubricity, radial force, Poisson’s ratio, and hardness). These updated testing methods subject the biomaterial to appropriate neurovascular physiological conditions, producing results relatable to the human condition. The results are analyzed and compared to previously tested human carotid vessel samples. This data can be used to “tune” the resulting 3D-prints, with optimal material properties, for improved in vitro vessel modeling, endovascular device deployment testing, and blood flow-related fatigue testing.
- The DAR study aims to create predictable in vitro and in vivo aneurysm rupture models using collagenase enzymes, that are known to weaken the tunica adventitia (outermost layer of blood vessels). Controlling the collagenase dose, imaging the vessels, and testing the vessels for mechanical properties may help us create vessel model analogs. The models can be connected to our comprehensive blood flow system, which simulates precise patient blood flows and pressures. Using novel 3D-printed biomaterials with predictable rupture characteristics may result in advanced models for testing future medical devices that can better prevent giant aneurysm ruptures in patients.
- This project investigates the influence of material phases and their volume fractions on the elastic behavior of mathematically-derived scaffolds, for use as a potential bone scaffold material. The scaffolds were 3D-printed with various multiphase materials in the same print. Variations in the elastic behavior confirmed that different layering patterns yield adjustable anisotropic material properties within the scaffolds. The preliminary data gathered from the bone scaffolds indicate that variations in stiffness properties can be altered to match physiological conditions such as bone tissue. Developing bone scaffolds that can closely approximate these variations in directional properties may dramatically improve bone regrowth, repair, and reorganization.
Computational blood flow modeling Accordion Closed
Dr. Tim Becker | Mechanical Engineering
The Bioengineering Devices Lab currently emphasizes two computational fluid dynamics (CFD) projects: 1) modeling of a balloon stent device for aneurysms, and 2) Modeling aspiration catheter flow effects for stroke treatment.
- The balloon-stent device will allow blood flow to perfuse through the parent artery while the neurointerventional surgeons treat aneurysms, which minimizes risk to the patient. Initial experiments with the simulated human condition – using a comprehensive blood flow system, attached to physiologically relevant 3D-printed artery – have shown that the balloon-stent device can provide sufficient blood flow during the entire treatment time. Computational Fluid Dynamics (CFD) has been used to verify the initial results, proving acceptable pressure drop and adequate blood flow past the balloon-stent device to avoid ischemic stroke.
- Mechanical thrombectomy, such as an aspiration catheter to remove blood clots in the brain, is now the standard of care for eligible patients with stroke symptoms. However, only half of patients who undergo aspiration achieve a favorable clinical outcome, in part due to current designs based on a limited understanding of the engineering principles of fluid flow and suction effects. Our new design maintains a small profile for accessibility to the patient blood clot, induces local flow arrest via a controllable and expandable catheter tip, and create a theoretical increase the aspiration force by up to 4x – improving clot incorporation and removal of the entire blood clot in the first attempt.
Medical device development and testing Accordion Closed
Dr. Tim Becker | Mechanical Engineering
The Bioengineering Devices Lab currently emphasizes four medical device development projects: 1) NeuroCURE liquid embolic for aneurysm treatment 2) balloon stent for aneurysm remodeling, 3) novel aspiration catheter for addressing gender disparity in ischemic stroke treatment, and 4) biocompatible polymer coating of current neurovascular devices:
- NeuroCURE® (Neurovascular Controlled Uniform Rapid Embolic) is an innovative liquid embolic designed to have little or no thrombus effects, high viscosity in liquid form, conformability for maximum aneurysm volume filling, minimal migration and particulation, non-adhesive gelling effects, relatively high mechanical strength, high elasticity, and biocompatibility in gel form. NeuroCURE is radiographically visible and fills to conform to an aneurysm sac while blood is displaced between the delivery catheter and the inflated balloon. Rapid injection (up to 2 ml/min) and a consistent gelation time (10 minutes) allows for fast, controllable delivery, resulting in complete aneurysm treatment.
- The balloon-stent device project goal is to develop, refine, and validate a new endovascular medical product that improves aneurysm neck protection during embolic device placement, while reducing blood flow obstruction in the parent artery and thus minimizing ischemic risk. The new device could expedite developments of novel treatments, without blocking the parent artery in the short-term or leaving an exposed metal device in the parent artery long-term. This supporting medical device that can improve the efficiency of aneurysm treatment, without significantly affecting downstream blood flow, drastically improving patient outcomes.
- 87% of stroke patients experience an ischemic stroke (blood clot). In order to save brain tissue and maximize long-term functional recovery, clot removal (thrombectomy) must be performed quickly. However, only half of patients who undergo mechanical thrombectomy achieve a favorable clinical outcome. Furthermore, women who are treated are less likely to fully recover, resulting in often worse functional outcomes compared to men. There is an unmet clinical need for a thrombectomy device with an adjustable tip design to improve clot-device contact (clot integration) and clot removal (clot digestion). This project investigates the disparity observed between men and women and focuses on developing and testing a novel aspiration catheter within benchtop vessel models that are representative of both men and women.
- Protein adsorption onto an implanted material is the human body’s first response to a foreign material. The types and quantities of adsorbed proteins on a foreign material dictate how cells respond. A novel polymer, based on NeuroCURE technology, has exhibited minimal inflammatory response when implanted, yet elicits rapid and controlled tissue growth and repair (neointimal formation – NI), as verified with histology. This novel polymer coating is being investigated to understand the basis of these compatibilities through analysis of protein adsorption (proteomics).
Medical imaging, analysis, and modeling Accordion Closed
Dr. Tim Becker | Mechanical Engineering
The Bioengineering Devices Lab currently emphasizes three medical imaging projects: 1) Patient brain vessel images converted to blood flow models, 2) NeuroCURE device visualization during implantation, and 3) voxel (3D volume) calculations of precise brain aneurysm sizes and volumes (morphology).
- We are taking actual patient computed tomography (CT) and magnetic resonance images (MRI) and creating benchtop 3D blood flow models with the anatomical accuracy, material properties, flow properties, and flow pressures of the actual patient. These multi-material, 3D-printed models can be used to improve patient diagnosis, help select the appropriate treatment device, and practice the treatment prior to the actual patient surgical intervention.
- Polymer devices, such as NeuroCURE, are made visible (radiopaque) under medical imaging by mixing in liquid contrast. However, the contrast often leaches out over time – reducing long-term visibility. As part of the lab’s FDA study, the long-term radiopacity of NeuroCURE is being assessed, for up to one year. NeuroCURE® is estimated to retain over 70% of its radiopacity after one year, which was validated in an animal model.
- We are determining the precise volume of a brain aneurysm from a series of CT and MRI images using a MathWorks® MATLAB algorithm. This algorithm has the potential to provide insight into unique aneurysm morphologies and improve cerebral aneurysm treatment. Currently, the area and volumetric calculations performed by the algorithm have proven to be accurate within <5%), error, whereas, with current estimation techniques, error was 20%.
3D-print biomaterials for human modeling Accordion Closed
Dr. Tim Becker | Mechanical Engineering
The Bioengineering Devices Lab currently emphasizes 3D-print biomaterials for human modeling: 1) 3D-printed biomaterials used in surgical simulations, and 2) A comprehensive blood flow modeling system.
- Our surgical models are in vitro vessel models for testing and validation of medical devices on the benchtop. They accurately simulate flow properties and surgical training without the need for direct human or animal interventions. New 3D-printed biomaterials, with a range of material properties in the same print, can create biomimetic models that accurately replicate the complex vessels of the brain. Preliminary data has allowed for the creation of several vessel models including an intracranial circle of Willis (CW) model along with several aneurysm vascular flow models to incorporate into a comprehensive flow modeling system
- A blood flow modeling system was created with the intention of mimicking the blood flow to the human brain (neurovasculature). A ViVitro Super pump is used to simulate human blood flow from the heart and pump CMC (a blood analog developed by the BDL) throughout the system. Solidworks is used in conjunction with a multi-material 3D printer to model, create, and test different blood vessel models. The benchtop flow system can operate at any blood pressure (typically set to 120/80 mmHg) and blood flow rate (typically 700 mL/min to the brain via the CW), and output the results in real-time.
Neuromuscular Mechanics of Human Movements Accordion Closed
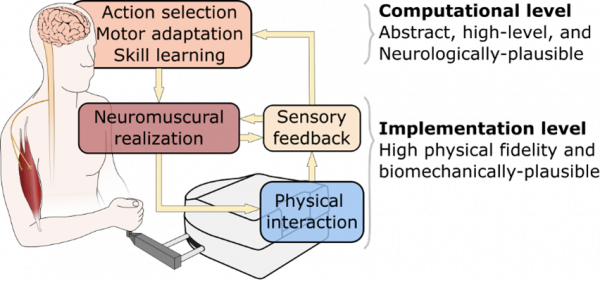
Lead: Reza Sharif Razavian
Keywords: human biomechanics, sensorimotor neuroscience, motor control, mathematical modeling
The sensorimotor apparatus that makes humans so dexterous is vastly complex. Our research philosophy is that the complexities of the human musculoskeletal system and its neural control mechanism are tightly interrelated, and one cannot be understood without considering the other. In our work, we aim to combine biomechanically plausible models of human movements with abstract neuroscientific theories to present multi-scale holistic models for human neuromuscular control. These holistic motor control models offer answers to both why- and how-questions in a unified framework. Low-level details such as the distinction between reflex-based and cortical contributions to muscle activities, and high-level “computational” aspects such as action selection and skill learning are studied together in this framework. In return, the model validation process will inspire new testable hypotheses and experimental paradigms to understand human movements better.
Precision Rehabilitation Engineering Accordion Closed

Lead: Reza Sharif Razavian
Keywords: rehabilitation robot, precision rehabilitation, human-aware control
A prominent technology in rehabilitation engineering is the use of robots to administer post-stroke exercises. This clinical practice, however, has been slow to progress despite its virtues, mostly because we do not know how a patient’s neuromuscular system responds to the intervention. Recent intelligent stroke rehabilitation robots can estimate a patient’s muscular activities during the exercise, thereby allowing the robot to optimally adapt to assist or resist the patient’s motion. Despite their abilities, current rehabilitation robots are still oblivious to the neuromuscular constraints of stroke survivors; electrophysiological studies indicate that patients lose part of their repertoire of neuromuscular modules for the brain to produce movement. In this research, we employ holistic motor control models in the robot’s controller to identify the patient’s neuromuscular constraints, and design exercise paradigms to push the patient out of their “neuromuscular comfort zone” to accelerate relearning of the lost motor function. Because of the heterogeneity of stroke survivors, “precision rehabilitation” is necessary—we need patient-specific exercises. Precision rehab necessities a realistic human movement model in the robot’s control algorithms.
Bio-Inspired Adaptive Exoskeleton Control Algorithms Accordion Closed

Lead: Zach Lerner
Keywords: exoskeleton, wearable robotics, control
Exoskeletons hold potential to augment walking ability, yet their use in free-living environments has been limited by the absence of practical and effective control strategies that can appropriately adapt to variable terrain, like stairs. To address this challenge, we are working on an analytical ankle joint moment estimation model using custom wearable sensors and developed an exoskeleton control scheme to adapt assistance proportional to the biological plantar-flexor moment in real-time.
Parallel-elastic Robotic Ankle Exoskeleton Accordion Closed

Lead: Zach Lerner
Keywords: exoskeleton, wearable robotics, actuator design
Actuation design that improves electromechanical efficiency and performance could increase the ability of untethered exoskeletons to provide meaningful assistance in free-living settings. We are working on designing a robotic ankle exoskeleton with a parallel elastic element in the form of a carbon fiber leaf spring to stored and returned energy in parallel to a cable-drive ankle joint during stance phase.
Real-world Exoskeleton Testing and Validation in Free-living Settings Accordion Closed
Lead: Zach Lerner
Keywords: exoskeleton, wearable robotics, biomechanics
The ability to readily complete challenging walking conditions is paramount to increasing and normalizing activities of daily living for ambulatory children and young adults with physical disabilities. Our overarching objective for this project is to augment mobility in challenging free-living settings for individuals with disabilities via ankle exoskeleton assistance. We are testing the use of the technology at home and in community settings.
Hip Exoskeleton Device Development Accordion Closed

Lead: Zach Lerner
Keywords: exoskeleton, wearable robotics, rehabilitation
The purpose of this project is to design and validate a novel autonomous hip exoskeleton with a user-adaptive control strategy capable of reducing the energy cost of level and incline walking in individuals with and without walking impairment.