Contact mechanical engineering
Managing faculty
Location
Shipping address
Mailing address
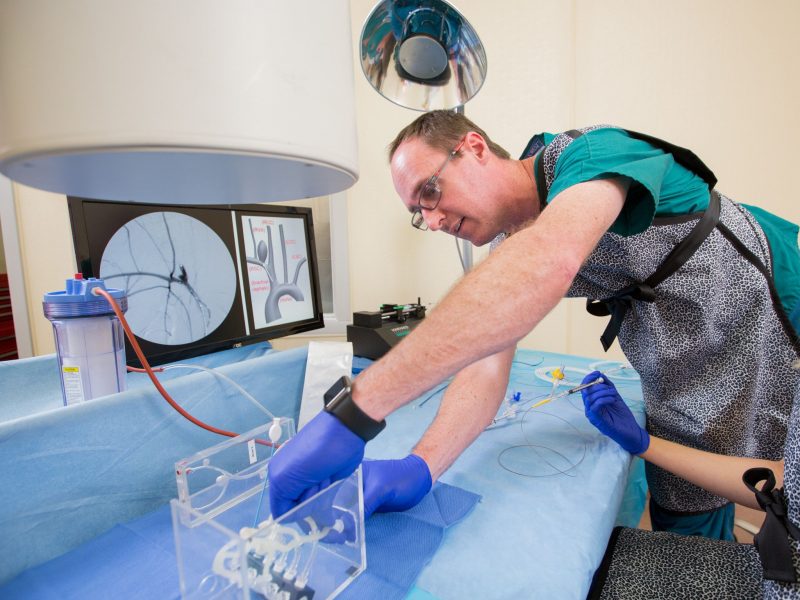
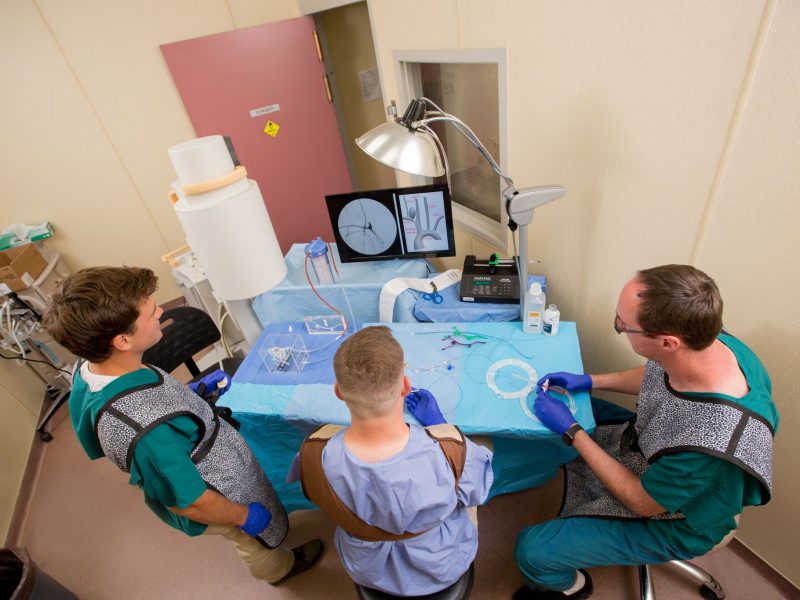
Bioengineering Devices Lab (BDL)
An NAU Mechanical Engineering Lab
The Bioengineering Devices Lab (BDL) is affiliated with the Bioengineering program and the Mechanical Engineering Department (ME) at Northern Arizona University.
The BDL provides opportunities to Ph.D., Masters, and undergraduate students interested in neurovascular devices such as new microcatheter designs, ischemic stroke systems, and innovative embolic agents for aneurysms (improving on coils, flow diverters, liquid embolics, etc). The lab conducts extensive in vitro cerebrovascular flow model development and simulates patient anatomies with innovative 3D-printed tissue-like biomaterials.

The lab is affiliated with Barrow Neurological Institute (BNI – Phoenix, AZ) and Northern Arizona Healthcare to collaborate on translational medical device research. Undergraduate and graduate researchers from the BDL actively collaborate with small start-ups and large-industry leaders in medical device development, published in internationally renowned journals, and attend annual conferences to present their work. Conferences regularly attended include BMES – Biomedical Engineering Society, Surfaces – Surfaces in Biomaterials Foundation, and SNIS – Society of NeuroInterventional Surgery.